In modern healthcare logistics, pharmaceutical distribution centers operate under some of the most demanding accuracy and compliance requirements in any industry. Unlike general e-commerce or retail fulfillment, a single error in medication sorting or shipment can have serious consequences for patient safety, regulatory compliance, and supply chain integrity.
As pharmaceutical networks grow more complex and globalized, distribution centers must process thousands to millions of SKUs with near-zero tolerance for mistakes. This is where automation technologies—especially cross-belt sorting systems—have become a transformative force.
Cross-belt sortation is not just about speed. It is about precision, traceability, redundancy, and real-time verification at every step of the fulfillment process. When properly integrated with advanced software systems such as warehouse management systems (WMS), serialization databases, and machine vision tracking, cross-belt technology can enable exceptionally high accuracy rates, approaching the theoretical goal of 100% traceability across every unit handled.
This article provides a deep technical and operational breakdown of how pharmaceutical distribution centers leverage cross-belt sortation systems to achieve highly reliable tracking accuracy, reduce human error, and meet strict regulatory expectations.

The Unique Challenges of Pharmaceutical Distribution
Pharmaceutical logistics differs fundamentally from traditional warehousing or retail distribution. The stakes are higher, the regulations are stricter, and the margin for error is significantly smaller.
Regulatory Requirements
Pharmaceutical distribution is governed by strict regulations such as serialization mandates, chain-of-custody documentation, and product verification requirements. In many regions, including the United States, regulations require end-to-end traceability of prescription medications.
This means every unit must be:
- Serialized with a unique identifier
- Tracked at every movement point
- Verified before shipment
- Logged in a compliant database
Any discrepancy can result in:
- Shipment rejection
- Regulatory penalties
- Product recalls
- Patient safety risks
High SKU Complexity
Pharmaceutical distribution centers often handle:
- Thousands of active SKUs
- Similar packaging formats
- Small size variations
- Temperature-sensitive products
- Controlled substances requiring special handling
This complexity increases the likelihood of mis-picks and mis-sorts without automation.
Time Sensitivity
Many pharmaceutical products are time-sensitive, including:
- Emergency medications
- Hospital supplies
- Temperature-controlled biologics
Distribution systems must balance speed and accuracy simultaneously, without compromising either.
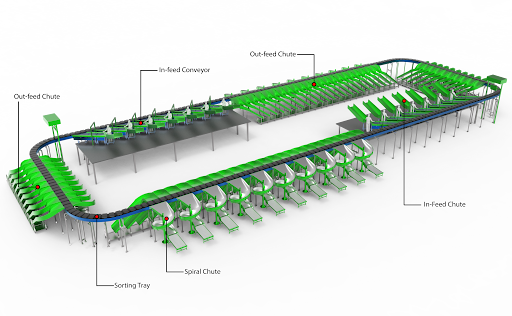
What Is Cross-Belt Sorting?
Cross-belt sorting is an automated material handling system designed for high-speed, high-precision sorting of individual items.
Basic Mechanism
A cross-belt sorter consists of a continuous loop conveyor system with small individual belt segments (called carriers). Each carrier has a mini conveyor belt that can move items sideways (perpendicular to the direction of travel).
The system operates as follows:
- Items are inducted onto the sorter
- Each item is scanned and identified
- The system assigns a destination chute
- The carrier transports the item
- The mini belt ejects the item into the correct chute
This process allows thousands of items per hour to be sorted with high precision.
Why It Is Ideal for Pharmaceuticals
Cross-belt systems excel in pharmaceutical environments because they provide:
- Gentle handling of delicate products
- High-speed processing
- Individual item tracking
- Precise destination control
- Minimal human intervention
The Core Concept: Accuracy Through Redundancy
Achieving near-perfect tracking accuracy is not the result of a single technology. Instead, it is the outcome of layered redundancy systems.
Cross-belt sorting systems integrate multiple verification points to ensure that every item is correctly identified, tracked, and routed.
Step 1: Item Identification at Induction
The accuracy journey begins when a pharmaceutical item enters the system.
Barcode and Data Capture
Each product is scanned using:
- High-resolution barcode scanners
- 2D data matrix readers
- RFID readers (in advanced systems)
This scan captures:
- National Drug Code (NDC)
- Serial number
- Lot number
- Expiration date
- Product description
Verification Against WMS
The scanned data is immediately validated against the warehouse management system.
If there is any mismatch, the item is diverted to an exception lane.
Step 2: Machine Vision Confirmation
Many advanced pharmaceutical distribution centers incorporate machine vision systems.
What Machine Vision Does
Machine vision cameras analyze:
- Label placement
- Packaging integrity
- Print quality
- Tampering signs
- Orientation
This ensures the item physically matches its digital identity.
Preventing Mislabeling Errors
Even if a barcode is readable, machine vision can detect:
- Incorrect labeling
- Duplicate serialization
- Damaged packaging
This second layer of validation significantly reduces downstream errors.
Step 3: Dynamic Routing Logic in Cross-Belt Systems
Once identified, each item is assigned a destination within the cross-belt system.
Algorithm-Based Sorting Decisions
The sorting engine uses real-time algorithms to determine:
- Optimal chute assignment
- Load balancing across lanes
- Priority shipment handling
- Temperature zone routing
Real-Time Decision Adjustments
If system conditions change, such as chute congestion or downstream delays, the system can dynamically reroute items without stopping operations.

Step 4: Controlled Item Transportation
After assignment, the item travels through the cross-belt conveyor.
Stable Handling Environment
Cross-belt systems are designed to ensure:
- Minimal vibration
- Stable item positioning
- Controlled acceleration
- Reduced drop risk
This is especially important for fragile pharmaceutical packaging such as vials, blister packs, and injectables.
Step 5: Precision Ejection Mechanism
At the correct destination point, the carrier belt activates and gently ejects the item into a designated chute.
Key Advantages of Cross-Belt Ejection
- Direct placement accuracy
- Reduced bouncing or misplacement
- High-speed throughput
- Minimal mechanical stress on products
This step is critical for maintaining system-wide accuracy.
Step 6: Secondary Verification at Sort Chutes
After sorting, additional verification systems confirm successful routing.
Post-Sort Scanning
At each chute, items may be re-scanned to verify:
- Correct destination
- Item identity match
- System confirmation logging
Weight and Dimension Checks
Some systems include sensors that verify:
- Expected weight range
- Package dimensions
This helps identify missing or incorrect items.
Step 7: Exception Handling Systems
No system is perfect, which is why exception management is essential.
What Triggers an Exception?
Items are flagged when:
- Barcode is unreadable
- Data mismatch occurs
- Physical damage is detected
- Routing conflict happens
Exception Handling Workflow
Instead of entering the main flow, exceptions are diverted to:
- Manual inspection stations
- Re-labeling areas
- Quality assurance review zones
This prevents faulty items from entering the supply chain.
The Role of Warehouse Management Systems (WMS)
A warehouse management system acts as the central intelligence layer.
Core Functions
WMS platforms manage:
- Inventory tracking
- Order processing
- Routing logic
- Compliance documentation
- Real-time system updates
Real-Time Data Synchronization
Every scan event updates the WMS instantly, ensuring:
- Full traceability
- Accurate inventory levels
- Regulatory compliance logs
Serialization and Track-and-Trace Integration
Serialization is critical in pharmaceutical logistics.
Unique Identification Per Unit
Each pharmaceutical unit is assigned a unique serial number, enabling:
- Individual tracking
- Anti-counterfeit protection
- Supply chain visibility
Regulatory Compliance Systems
Systems integrate with national and global databases to ensure:
- Product authenticity
- Chain-of-custody validation
- Regulatory reporting compliance
How Cross-Belt Systems Reduce Human Error
One of the primary advantages of automation is error reduction.
Eliminating Manual Sorting Risks
Human sorting errors can include:
- Misreading labels
- Incorrect item placement
- Fatigue-related mistakes
Cross-belt systems eliminate most manual handling steps.
Controlled Human Interaction Points
Humans are still involved, but only in:
- Exception handling
- System supervision
- Quality control audits
This minimizes risk exposure.
Achieving Near-100% Accuracy: The Reality
While no system can guarantee absolute perfection in all conditions, modern cross-belt pharmaceutical systems achieve extremely high accuracy rates due to layered safeguards.
Key Accuracy Drivers
Accuracy is achieved through:
- Multi-layer scanning
- Machine vision verification
- Real-time WMS validation
- Physical routing control
- Exception diversion systems
Why Redundancy Works
If one system layer fails, others compensate.
For example:
- If barcode scanning fails, RFID may succeed
- If routing misfires, chute verification detects it
- If data mismatch occurs, system halts the item
This redundancy is the foundation of high-integrity logistics.
Temperature-Controlled Pharmaceutical Distribution
Many pharmaceutical products require controlled environments.
Cold Chain Integration
Cross-belt systems can operate alongside:
- Refrigerated zones
- Freezer storage areas
- Temperature-monitored conveyors
Monitoring and Alerts
Sensors continuously track:
- Ambient temperature
- Exposure duration
- Handling conditions
If thresholds are exceeded, alerts are triggered.
Scalability of Cross-Belt Systems
Modern distribution centers must scale efficiently.
Modular Expansion
Cross-belt systems are often modular, allowing:
- Additional lanes
- Increased throughput capacity
- Flexible facility design
Handling Peak Demand
During high-demand periods, such as flu season, systems can process dramatically higher volumes without sacrificing accuracy.
Data Analytics and Continuous Improvement
Modern logistics systems are highly data-driven.
Performance Metrics
Operators analyze:
- Sorting accuracy rates
- Throughput speed
- Exception frequency
- Downtime events
Machine Learning Optimization
Advanced systems use predictive analytics to:
- Optimize routing efficiency
- Reduce congestion
- Improve system uptime
Security and Anti-Counterfeit Protection
Pharmaceutical integrity is critical.
Preventing Counterfeit Entry
Cross-belt systems help ensure:
- Verified product sourcing
- Authentic serialization checks
- Controlled chain-of-custody tracking
Tamper Detection
Machine vision systems can detect signs of:
- Packaging tampering
- Label inconsistencies
- Unauthorized modifications
Future Innovations in Pharmaceutical Sorting
The future of pharmaceutical logistics will likely include even greater automation and intelligence.
AI-Driven Sorting Optimization
Artificial intelligence may further improve:
- Routing decisions
- Predictive maintenance
- Error detection
Fully Autonomous Distribution Centers
Next-generation facilities may operate with minimal human intervention, relying on:
- Robotics
- Autonomous conveyors
- Intelligent scanning systems
Blockchain-Based Tracking
Blockchain may be used to enhance:
- Supply chain transparency
- Immutable tracking records
- Cross-organizational verification
Challenges and Limitations
Despite advances, challenges remain.
System Complexity
Highly automated systems require:
- Skilled maintenance teams
- Continuous monitoring
- Software updates
Initial Investment Costs
Cross-belt systems require significant upfront capital investment.
However, long-term efficiency gains often offset these costs.
Final Thoughts
Cross-belt sorting systems represent one of the most advanced technologies in pharmaceutical distribution today. By combining high-speed mechanical sorting with layered digital verification systems, machine vision, real-time warehouse intelligence, and strict regulatory integration, these systems achieve exceptionally high levels of tracking accuracy.
The key to approaching near-perfect accuracy is not relying on a single technology, but rather building a multi-layered ecosystem where every step validates the previous one.
From induction scanning to final chute verification, every stage contributes to a closed-loop system designed to eliminate errors before they can propagate.
As pharmaceutical supply chains continue to grow in complexity, cross-belt sorting will remain a foundational technology for ensuring safety, reliability, and trust in global medicine distribution networks.